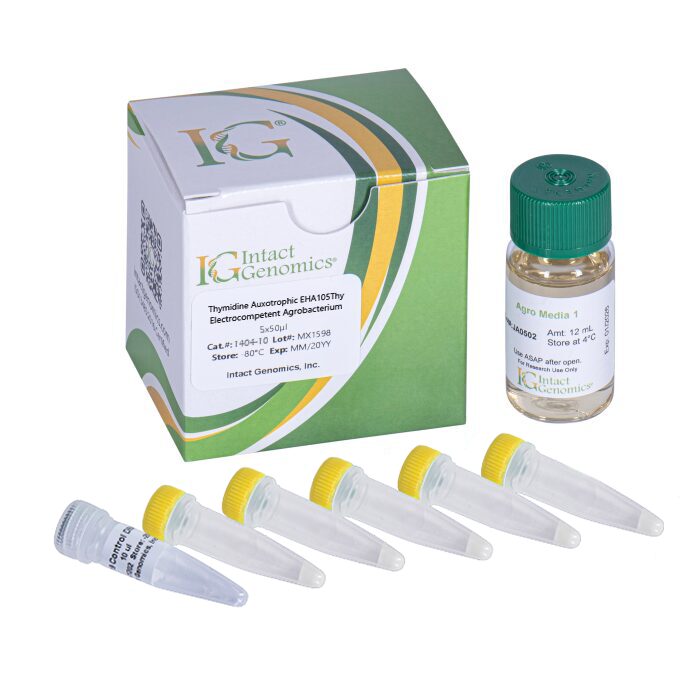
ig® Thymidine-Auxotrophic EHA105 Agro cells includes modifications so that they will not grow unless thymidine is added to Minimal medium. This prevents the bacteria from overgrowing plant tissues.
Thymidine Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells
Price range: $376.00 through $695.00
Description
Intact Genomics (ig®) Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells include modifications so that they will not grow unless 50 mg/L of thymidine is added to Minimal medium. This prevents the bacteria from overgrowing plant tissues when used for plant transformation. Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells are optimized for the highest transformation efficiencies. The EHA105Thy strain is useful for transgenic operations of rice, tobacco and other plants. EHA105Thy– thyA knockout mutant derived from EHA105, itself a derivative of A281 (A136/pTiBo542).
Custom Aliquots Available
Benefits:
- Thymidine Auxotrophic
- Enables development of more efficient transformation systems
- Reduced bacterial overgrowth during co-cultivation
- Decreased need for antibiotics
Specifications:
Competent cell type: Electrocompetent
Species: A. tumefaciens
Strain: EHA105Thy
Format: Tubes
Transformation efficiency: ≥ 1 x 103 cfu/µg pCAMBIA1391z DNA
Shipping condition: Dry ice
Product Components:
- ig® Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells
- DNA (pCAMBIA1391z, 500 pg/µl)
- Recovery medium
Storage:
- ig® EHA105Thy Agrobacterium ElectroCompetent Cells: -80 °C
- pCAMBIA1391z control DNA: -20 °C
- Recovery medium: 4 °C
Quality Control:
Transformation efficiency is tested by using the pCAMBIA1391z control DNA supplied with the kit and using the protocol in this manual. Transformation efficiency should be ≥1 x 103 CFU/µg pCAMBIA1391z DNA. Untransformed cells are tested for appropriate antibiotic sensitivity.
General Guidelines:
Follow these guidelines when using ig® Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells:
- Handle competent cells gently as they are highly sensitive to changes in temperature or mechanical lysis caused by pipetting.
- Thaw competent cells on ice and transform cells immediately following thawing. After adding DNA, mix by tapping the tube gently. Do not mix cells by pipetting or vortexing.
Calculation of Transformation Efficiency:
Transformation Efficiency (TE) is defined as the number of colony forming units (cfu) produced by transforming 1µg of plasmid into a given volume of competent cells.
TE = Colonies/µg/Plated
Transform 1 µl of (500 pg/µl) pCAMBIA1391z control plasmid into 25 µl of cells, add 974 µl of Recovery Medium. Recover for 3 hours and plate 100 µl. Count the colonies on the plate in two days. If you count 500 colonies, the TE is calculated as follows:
Colonies = 500
µg of DNA = 0.0005
Dilution = 100/1000 = 0.1
TE = 500/.0005/.1 = 1×107
1404-10 1404-30
Additional information
| μl | 5×50μl, 15×50μl, 6×100μl, 12×100μl |
|---|
References
Aliu E., Azanu MK., Wang K., Lee K. Generation of thymidine auxotrophic Agrobacterium tumefaciens strains for plant transformation https://www.biorxiv.org/content/10.1101/2020.08.21.261941v1.full.pdf
Additional Information:
The auxotrophic Agrobacterium tumefaciens strains were developed with support by National Science Foundation Plant Genome Research Program Grant 1725122 and 1917138 to K.W., by the Iowa State University Interdepartmental Plant Biology Major fellowship to EA and MA, by the USDA NIFA Hatch project #IOW04341 and by State of Iowa funds and by the Crop Bioengineering Center of Iowa State University.
Technical Support
Intact Genomics (IG®) is dedicated to customer satisfaction regarding the use of our products for your research needs. We test our products thoroughly to ensure they conform to the highest quality standards and provide excellent results when following the protocol’s specifications. Please follow the protocol information provided in this manual carefully and contact our customer and technical support team with any questions or comments you may have regarding this or our other products.
IG Technical Support
Email: sales@intactgenomics.com
Phone: (314) 942-3655 | Toll free: 855-835-7172
Legal Notices
Intact Genomics owns the following registered trademarks granted by the United States Patent and Trademark Office (USPTO): Intact Genomics®, IG®, ig®, igTherapeutics®, FastAmp®, i7®, DirectPlate® .
All technology protocols discussed within this manual are assumed proprietary to IG’s products, and if registered, are protected by US patent and trademark laws.
Notice to Buyer – Limited License
Buyer agrees by purchasing the product that it’s intended use is for research purposes only, not for human or diagnostic use. Purchase of this material conveys to buyer the non-transferable right to use the material purchased in research conducted by buyer, whether for teaching, non-commercial or commercial research purposes. Buyer may not sell or otherwise transfer these materials, its components, or unmodified descendants to a third party. Use of these products maybe covered by IG’s patents or trademarks.
Products are understood by buyer to be experimental in nature and may have hazardous properties. Unless prohibited by law, buyer assumes all liability for claims or damages against it by third parties that relate to or arise from the use, storage, or disposal of the purchased materials.
Buyer agrees to use the purchased materials in full compliance with applicable law and regulations. Intact Genomics will not be held liable for activities outside of itself and the products intended use.
For more information or to express legal concerns relating to patent and trademarks, please contact us at: info@intactgenomics.com or sales@intactgenomics.com
Transformation Protocol:
Use this procedure to transform ig® Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent Cells. Do not use these cells for chemically transformation.
- Place sterile cuvettes and microcentrifuge tubes on ice.
- Remove ig® Thymidine-Auxotrophic EHA105Thy Agrobacterium ElectroCompetent cells from the -80 °C freezer and thaw completely on wet ice (10-15 minutes).
- Aliquot 1 µl (10pg -1 µg) of DNA to the chilled microcentrifuge tubes on ice.
- When the cells are thawed, add 25 μl of cells to each DNA tube on ice and mix gently by tapping 4-5 times.
For the pCAMBIA1391z control, add 1 µl of (500 pg/µl) DNA to the 25 µl of cells on ice. Mix well by tapping.
Do not pipette up and down or vortex to mix, this can harm cells and decrease transformation efficiency. - Pipette 26 µl of the cell/DNA mixture into a chilled electroporation cuvette without introducing bubbles. Quickly flick the cuvette downward with your wrist to deposit the cells across the bottom of the well and
then electroporate. - Immediately add 974 µl of Recovery Medium with 50 mg/L of thymidine or any other medium of choice to the cuvette, pipette up and down three times to re-suspend the cells. Transfer the cells and Recovery Medium to a 15ml of culture tube.
- Incubate tubes at 30 °C for 3 hours at 200 RPM.
- Spread 20-200 μl cells onto a pre-warmed selective plate. For the pCAMBIA1391z control, you may plate 100 μl of undiluted transformation mix onto a YT plate containing 50 μg/ml of thymidine, 15 μg/ml rifampicin and 50 μg/ml kanamycin. Use sterilized spreader or autoclaved ColiRoller™ plating beads to spread evenly.
- Incubate the plates for 2 – 3 days at 30 °C.
Electroporation Settings:
Mode: Exponential protocol
Voltage (V): 1,800 V
Capacitance: 25 uFD
Resistance: 200 Ohms
Cuvette: 1 mm