Description
Endonuclease IV (Nfo) from Escherichia coli is a 32-kD metalloprotein that aids in the repair of damaged DNA. The enzyme functions both as an apurinic/apyrimidinic nuclease (1) and as a 3′-terminal di-esterase (1-4). Its 3′-terminal di-esterase activity is important in the repair of DNA strand breaks generated by oxidation and ionic radiation (2, 3). In such events, the strand breaks terminate with either a 3′ phosphate or a deoxyribose fragment, preventing repair by DNA polymerase I or DNA ligase. Endonuclease IV removes the blocking groups, leaving a free 3′-hydroxyl terminus. This enzyme does not have detectable associated exonuclease or DNA N-glycosylase activity (1).
Applications
• Single cell gel electrophoresis (Comet assay) (5, 6)
• Alkaline elution (7)
• Alkaline unwinding (8)
Protein purity
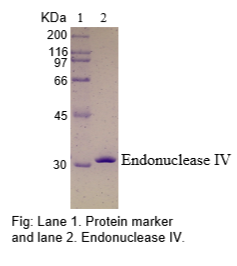
The physical purity of this enzyme is ≥99% as assessed by SDS-PAGE with Coomassie® blue staining (see figure below).
Product Source
E. coli BL21 (DE3) strain expressing E. coli Endonuclease IV gene.
Product Includes
• Endonuclease IV (Nfo)
• 10x Endonuclease IV reaction buffer
1x Endonuclease IV reaction buffer
50 mM Tris-HCl
10 mM MgCl2 1 mM DTT 100 mM KCl (pH 7.9 @ 25°C)
Storage Buffer
50 mM Tris-HCl, 50 mM KCl, 1 mM DTT, 0.1 mM EDTA, 50% Glycerol, pH 7.5 @ 25ºC
Storage Temperature
-20ºC
Heat inactivation
85°C for 20 min
Unit Definition
One unit is defined as the amount of enzyme required to cleave 1 pmol of a 50-mer oligonucleotide duplex containing a single AP site in a total reaction volume of 10 μl in 1 hour at 37°C.
Quality Control assays
Endonuclease IV is free from detectable contaminating nuclease activities.
References
1. Ljungquist, S. (1977) J. Biol. Chem. 252, 2808.
2. Demple, B. et al., (1986) Proc. Natl. Acad. Sci. USA 83, 7731.
3. Levin, J.D. et al., (1988) J. Biol. Chem. 263, 8066.
4. Levin, J.D. et al., (1991) J. Biol. Chem. 266, 22893.
5. Singh, N. et al. (1961). Experimental Cell Research. 175, 184-191.
3422, 3425